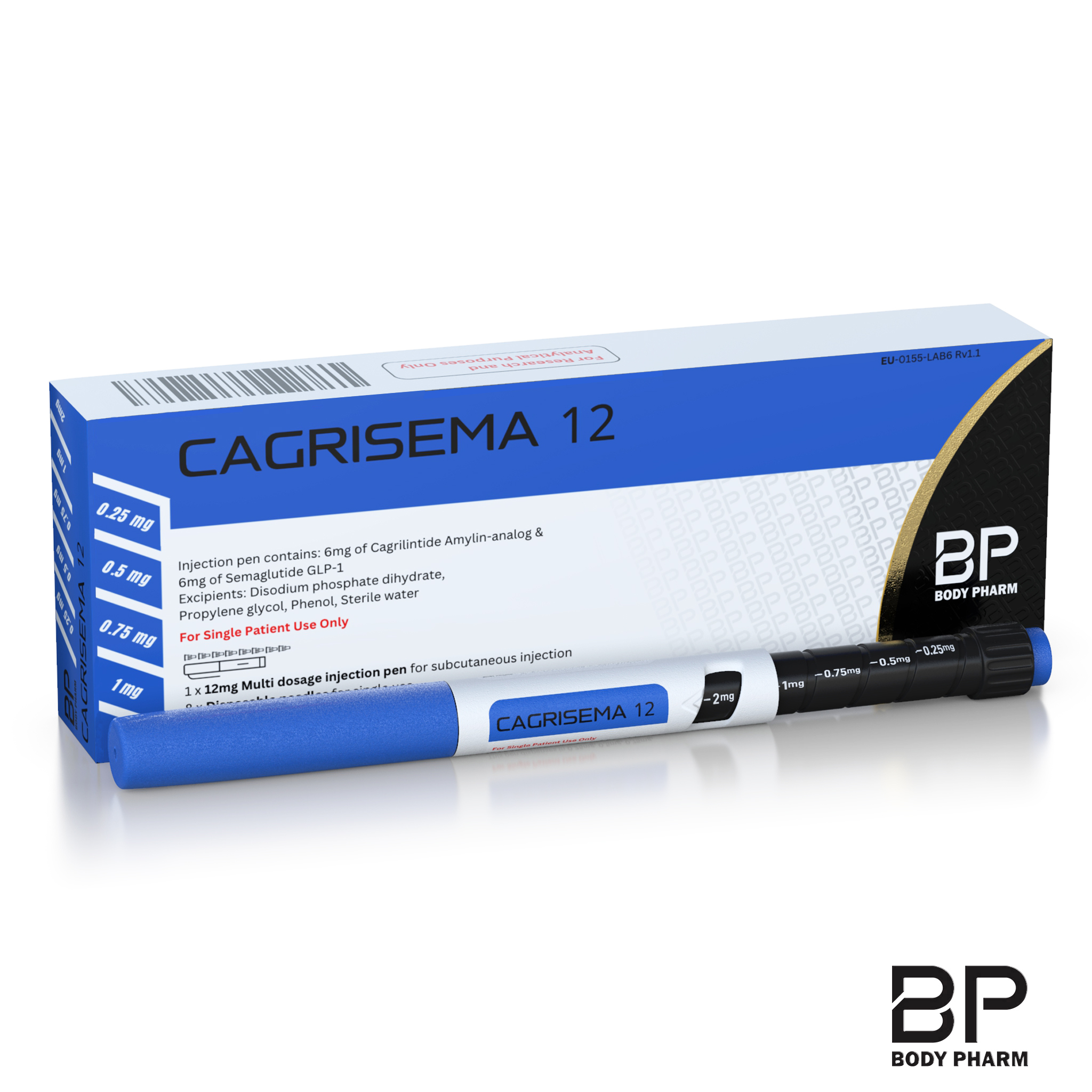
Body Pharm Cagrisema 12 pen
R2500,00
25 in stock
Description
Body Pharm Cagrisema 12 Pen: Dosage, Benefits & How to Buy
The Body Pharm Cagrisema 12 pen is a prefilled multi-dose injection device combining cagrilintide (a long-acting amylin analogue — a synthetic version of the pancreatic hormone amylin) with semaglutide (a Glucagon-Like Peptide-1 (GLP-1) receptor agonist) at a 12 mg total peptide load, sold in South Africa for weight loss and glycaemic (blood-sugar) support. In Novo Nordisk’s Phase 2 SCALE-NEXT trial, the same combination delivered roughly 15.6% mean body weight reduction at 32 weeks — nearly double the ~8.0% achieved by semaglutide alone (The Lancet, 202301548-3/fulltext)). This guide covers the legal realities of buying a product that is not registered with the South African Health Products Regulatory Authority (SAHPRA) (SAHPRA), how to escalate dosage, expected benefits, and side effects.
Key Takeaways
- What it is: A once-weekly subcutaneous pen combining 6 mg semaglutide and 6 mg cagrilintide for weight loss and blood-sugar control.
- Efficacy: ~15.6% mean weight loss at 32 weeks in Phase 2 trials — roughly double semaglutide alone.
- Dosing: 16-week titration from 0.25 mg to 2 mg once weekly to limit gastrointestinal (GI) side effects.
- Risks: Nausea (~40%), vomiting, diarrhoea; rare pancreatitis and gallbladder events; contraindicated in medullary thyroid carcinoma history.
- Legal status: Semaglutide is a Schedule 4 medicine in South Africa; Cagrisema itself is not SAHPRA-registered.
- Price: R2,500 per pen via Beskinny.store, with cold-chain delivery nationwide.
By the end of this article, you will understand (1) how the dual semaglutide–cagrilintide mechanism works, (2) how to titrate and inject the pen safely, and (3) the regulatory and clinical considerations before buying.
What Is the Body Pharm Cagrisema 12 Pen?
The Body Pharm Cagrisema 12 pen is a prefilled multi-dose injection device containing 6 mg semaglutide + 6 mg cagrilintide (12 mg total peptide load) in a single cartridge. South African compounding brand Body Pharm manufactures the pen for once-weekly subcutaneous use. The device targets two appetite-regulating pathways at once — GLP-1 and amylin — for compounded weight-loss and glycaemic effects.
Unlike single-agent pens such as the HD Semaglutide 10 Pen, Cagrisema co-delivers cagrilintide, a long-acting amylin analogue with a ~7-day half-life that matches semaglutide’s 165–184 hour half-life. The matched half-lives synchronise once-weekly dosing (The Lancet, 202101751-7/fulltext)). This dual mechanism drove the 15.6% mean weight reduction observed in SCALE-NEXT versus 8.0% for semaglutide alone.
Some readers may wonder whether a compounded combination is as reliable as branded single-agent pens. Body Pharm pens are not produced under Novo Nordisk’s regulatory file, so quality assurance depends on the compounder and supplier — a factor we expand on in the eligibility and storage sections below.
Pen Format and Dose Increments
The pen is a click-dial device that delivers five selectable increments per injection: 0.25 mg, 0.5 mg, 0.75 mg, 1 mg, and 2 mg, as confirmed by South African retailers including myantidote.co.za. This range covers the full titration ladder from starter dose through maintenance. A single 12 mg pen typically lasts 4–8 weeks depending on your stage of escalation.
For users who progress past Cagrisema’s ceiling, the Body Pharm Tirzepatide 30 Pen offers a higher-potency alternative on the same delivery format.
Semaglutide + Cagrilintide: How the Dual Mechanism Works
Cagrisema activates two appetite-regulating hormone systems at once — GLP-1 (via semaglutide) and amylin (via cagrilintide). The two peptides suppress hunger through separate hypothalamic satiety pathways, slow gastric emptying via complementary mechanisms, and blunt postprandial (post-meal) glucose spikes more effectively than either agent alone. Because the receptors are non-overlapping, the effects add together rather than duplicate. This dual action produced ~15.6% mean body-weight reduction versus ~8.0% for semaglutide monotherapy in the Phase 2 SCALE-NEXT trial (The Lancet, 202301548-3/fulltext)).
A common concern is whether activating two systems doubles the side-effect burden. In practice, GI side effects rise modestly (see Side Effects section) but serious adverse events remain in line with the GLP-1 class.
What Each Peptide Does
Semaglutide is a GLP-1 receptor agonist that mimics the incretin hormone glucagon-like peptide-1. The drug stimulates glucose-dependent insulin secretion (only when blood glucose is elevated, minimising hypoglycaemia risk), suppresses glucagon release from pancreatic alpha cells, reduces hepatic (liver) glucose output, and delays gastric emptying — extending satiety after meals (FDA Wegovy Prescribing Information).
Cagrilintide is a long-acting analogue of amylin, a hormone co-secreted with insulin by pancreatic beta cells. The peptide binds amylin and calcitonin receptors in the area postrema and hypothalamus — anatomically distinct from GLP-1 receptor sites — driving satiety through a parallel neural circuit. Cagrilintide further slows gastric emptying and suppresses postprandial glucagon, compounding semaglutide’s glycaemic effects (Enebo et al., The Lancet 202101751-7/fulltext)).
Side-by-Side Mechanism Comparison
| Action | Semaglutide (GLP-1 agonist) | Cagrilintide (amylin analogue) |
|---|---|---|
| Primary satiety pathway | GLP-1 receptors in hypothalamus & brainstem | Amylin/calcitonin receptors in area postrema |
| Insulin secretion | Stimulates (glucose-dependent) | Modulates indirectly via glucagon suppression |
| Glucagon | Suppresses | Suppresses (postprandial) |
| Gastric emptying | Significantly delayed | Further delayed (additive) |
| Hepatic glucose output | Reduced | Indirect reduction |
| Half-life | 165–184 hours (~7 days) | ~168 hours (~7 days) |
Why the Combination Outperforms Monotherapy
Because the two peptides engage non-overlapping receptors, their appetite-suppressive effects add together rather than duplicate. Single-agent pens like the HD Semaglutide 10 Pen activate only the GLP-1 axis, leaving amylin signalling untouched. Novo Nordisk’s ongoing REDEFINE Phase 3 programme (NCT05567796, NCT05394519) is testing whether the SCALE-NEXT effect size holds at 68 weeks, with readouts continuing through 2025 (ClinicalTrials.gov). For users seeking even higher efficacy, the Body Pharm Tirzepatide 30 Pen offers a GLP-1/Glucose-dependent Insulinotropic Polypeptide (GIP) dual-agonist alternative — a useful contrast for anyone evaluating a GLP-1/amylin combination against newer multi-agonist options.
Key Benefits of Cagrisema for Weight Loss & Blood Sugar
Cagrisema delivers six measurable advantages over single-agent GLP-1 therapy: greater absolute weight loss, day-long appetite control, tighter postprandial glucose, reduced food cravings, once-weekly dosing, and broad applicability across obesity (Body Mass Index (BMI) ≥30), type 2 diabetes (T2D), and metabolic syndrome — all driven by the additive pharmacology of cagrilintide and semaglutide acting on non-overlapping receptor systems.
Some readers may question whether these benefits translate outside controlled trials. The trial populations were closely supervised, so real-world results depend on titration discipline and clinician follow-up.
- Superior weight reduction vs. GLP-1 monotherapy — Phase 2 SCALE-NEXT data showed ~15.6% mean body weight loss at 32 weeks on cagrilintide 2.4 mg + semaglutide 2.4 mg, compared with ~8.0% on semaglutide monotherapy and ~5.1% on cagrilintide alone (Lau et al., The Lancet 202301548-3/fulltext)). Buyers comparing efficacy tiers can contrast this result against the HD Semaglutide 10 Pen.
- Sustained appetite suppression across the day — Amylin signalling in the area postrema and nucleus tractus solitarius prolongs meal-to-meal satiety, complementing semaglutide’s hypothalamic effect (Boyle et al., Physiology & Behavior 2018).
- Improved postprandial glucose control — Cagrilintide suppresses postprandial glucagon and further slows gastric emptying, flattening post-meal glucose excursions on top of semaglutide’s glucose-dependent insulin secretion (Enebo et al., The Lancet 202101751-7/fulltext)).
- Reduced cravings and emotional eating — Central amylin receptors modulate dopaminergic reward pathways linked to hedonic food intake — an axis untouched by GLP-1 agonists alone (Mietlicki-Baase, Physiology & Behavior 2016).
- Once-weekly injection convenience — Both peptides have ~7-day half-lives via albumin-bound fatty-acid acylation, enabling synchronised weekly dosing and steady-state plasma levels by week 4–5 (FDA Wegovy Label).
- Suitable for obesity, type 2 diabetes, and metabolic syndrome — REDEFINE 1 (obesity) and REDEFINE 2 (T2D + overweight) confirm clinical relevance across both populations. Users seeking even higher weight-loss ceilings may consider the Body Pharm Tirzepatide 30 Pen as a GLP-1/GIP alternative (ClinicalTrials.gov NCT05394519).
Cagrisema 12 Dosage & Escalation Schedule
The Body Pharm Cagrisema 12 dosage protocol follows a 16-week titration. The schedule starts at 0.25 mg once weekly and escalates every 4 weeks to a 2 mg maintenance dose, mirroring the FDA-approved semaglutide protocol used in Ozempic and Wegovy. Slow upward titration matters because the gradual ramp lets GLP-1 and amylin receptors adapt, sharply reducing the nausea, vomiting and diarrhoea seen with rapid initiation (FDA Wegovy Label).
A frequent objection is that 16 weeks feels slow when the goal is rapid weight loss. However, skipping steps consistently predicts treatment dropout from GI intolerance, so the slower path usually delivers better long-term results.
Week-by-Week Escalation Table
| Phase | Weeks | Dose (once weekly) | Purpose |
|---|---|---|---|
| Initiation | 1–4 | 0.25 mg | Receptor adaptation; minimise GI effects |
| Step-up 1 | 5–8 | 0.5 mg | Begin appetite suppression |
| Step-up 2 | 9–12 | 0.75 mg | Progressive satiety, early weight loss |
| Step-up 3 | 13–16 | 1.0 mg | Approach therapeutic threshold |
| Maintenance | 17+ | 2.0 mg | Full REDEFINE-equivalent dose |
The pen’s calibrated dial selects each step precisely, so you do not need to withdraw from a syringe or convert units. Skipping a titration step is the single biggest predictor of treatment discontinuation due to GI intolerance (Wilding et al., NEJM 2021).
Injection Technique
Inject subcutaneously into the abdomen (at least 5 cm from the navel), the front of the thigh, or the back of the upper arm. Rotate the site each week to prevent lipohypertrophy — a hardened lump that impairs absorption. Hold the pen needle against the skin for 6 seconds after the dose clicks complete to ensure full delivery. Pick one fixed day of the week (e.g. every Sunday morning) and inject within a ±48-hour window of that anchor day. When you miss the window by more than 48 hours, skip that week and resume on schedule.
Prescription Status & Practitioner Supervision
Semaglutide is a Schedule 4 medicine under SAHPRA in South Africa, requiring a valid prescription from a registered medical practitioner (Medicines and Related Substances Act 101 of 1965). A clinician should baseline your Haemoglobin A1c (HbA1c), renal function, and thyroid history before initiation, and review you at weeks 4, 12 and 24. Should 2 mg prove intolerable, dose reduction to 1 mg is clinically valid and still produces meaningful weight loss.
Who Is Cagrisema 12 Suitable For?
Cagrisema 12 best suits adults with a BMI ≥30 kg/m² (obesity), or BMI ≥27 kg/m² alongside a weight-related comorbidity such as type 2 diabetes, hypertension, dyslipidaemia, or obstructive sleep apnoea — the same threshold criteria used in the REDEFINE 1 and REDEFINE 2 Phase 3 trials (ClinicalTrials.gov NCT05567796). The pen also suits patients who have plateaued on GLP-1 monotherapy and want the additive amylin effect; SCALE-NEXT showed ~15.6% weight loss at 32 weeks on the combination versus ~8.0% on semaglutide alone (Lau et al., Lancet 202301548-3/fulltext)).
Have you already tried the HD Semaglutide 10 Pen without reaching your target? This semaglutide–cagrilintide combination is a logical step up. For patients who need stronger glycaemic control, the Body Pharm Tirzepatide 30 Pen is the next tier.
A reasonable concern is whether the combination fits patients with mild metabolic issues. For BMI under 27 with no comorbidity, the risk-benefit balance generally does not favour Cagrisema; lifestyle interventions or single-agent therapy are usually more appropriate.
Absolute Contraindications
Do not use Cagrisema if you have a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2) — a class-wide FDA black-box warning for all GLP-1 receptor agonists (Wegovy US Prescribing Information). Other contraindications include prior pancreatitis, pregnancy or breastfeeding, severe renal impairment (estimated Glomerular Filtration Rate (eGFR) <30 mL/min/1.73m²), and known hypersensitivity to semaglutide or cagrilintide.
Use With Caution
Combining Cagrisema with insulin or sulfonylureas significantly increases hypoglycaemia risk; usually you must reduce the dose of the background agent (Ozempic Prescribing Information §7). A clinician should evaluate patients with gastroparesis, severe Gastro-Oesophageal Reflux Disease (GORD), or active gallbladder disease individually. Before your first dose, request baseline HbA1c, eGFR, lipase, and a thyroid history review — these tests are non-negotiable safety screens.
Potential Side Effects & How to Manage Them
Most Cagrisema side effects are gastrointestinal, dose-dependent, and resolve within 4–8 weeks as your gut adapts. The dual mechanism does increase nausea over semaglutide alone, but serious adverse events remain rare when you respect the titration schedule.
A common worry is whether GI side effects make the pen impractical for working adults. In trials, however, most patients tolerated symptoms with simple meal-timing changes and continued therapy.
Common Effects (>10% of users)
In the SCALE-NEXT Phase 2 trial, nausea affected approximately 40% of combination-arm participants versus ~30% on semaglutide alone, with the vast majority graded mild-to-moderate (Lau et al., Lancet 202301548-3/fulltext)). Vomiting, diarrhoea, constipation, and transient injection-site redness or itching round out the high-frequency list. These effects peak in the 1–2 weeks following each dose escalation and taper sharply thereafter.
Less Common Effects
Headache, fatigue, dizziness, and reduced appetite-driven mild dehydration occur in 5–10% of users. Heart rate rises modestly (2–4 beats per minute), consistent with the GLP-1 class profile (Wegovy Prescribing Information).
Rare but Serious — Stop and Seek Care
Discontinue immediately and contact a doctor for: severe, persistent abdominal pain radiating to the back (possible pancreatitis), right-upper-quadrant pain with nausea or jaundice (gallbladder disease — incidence ~2.6% in semaglutide trials), or hypoglycaemia symptoms when you use insulin or sulfonylureas concurrently (Ozempic Prescribing Information).
Practical Management
- Inject after a light meal for the first 4 weeks to blunt nausea.
- Hydrate aggressively — drink 2.5–3 L water daily to offset reduced thirst signalling.
- Eat smaller, lower-fat meals; high-fat foods worsen delayed gastric emptying.
- Hold the next escalation when current symptoms remain moderate — staying at a tolerated dose for an extra week is safer than pushing through.
- Report persistent vomiting beyond 48 hours to your prescriber; dehydration can precipitate acute kidney injury.
When GI tolerability remains the dealbreaker, the milder HD Semaglutide 10 Pen is a gentler entry point before considering combination therapy.
Cagrisema vs. Semaglutide Alone: Is the Combination Worth It?
For patients plateauing on GLP-1 monotherapy or needing tighter dual metabolic control, yes — Cagrisema’s combination of cagrilintide and semaglutide delivers roughly double the weight loss of semaglutide alone (~15.6% vs ~8.0% at 32 weeks in SCALE-NEXT). The pen adds amylin-driven satiety on top of GLP-1 action, making Cagrisema the stronger choice for obesity treatment when single-agent results stall (The Lancet, 202301548-3/fulltext)).
A reasonable concern is whether the higher cost (R2,500 per pen vs entry-level semaglutide) justifies the gain. For patients still losing weight on semaglutide alone, the answer is usually no; the upgrade matters most after a plateau.
Side-by-Side Comparison
| Attribute | Body Pharm Cagrisema 12 Pen | HD Semaglutide 10 Pen | Body Pharm Tirzepatide 30 Pen |
|---|---|---|---|
| Active ingredients | Cagrilintide + semaglutide (amylin + GLP-1) | Semaglutide (GLP-1 only) | Tirzepatide (GIP + GLP-1) |
| Mean weight loss | ~15.6% at 32 weeks (SCALE-NEXT) | ~8.0% at 32 weeks (Lancet 202301548-3/fulltext)) | ~20.9% at 72 weeks (SURMOUNT-1, NEJM 2022) |
| HbA1c reduction (T2D) | ~1.8 percentage points | ~1.4 percentage points (Ozempic PI) | ~2.0–2.4 percentage points (SURPASS) |
| Appetite suppression | Deep — dual receptor (satiety + delayed gastric emptying) | Moderate — GLP-1 mediated | Deep — dual incretin |
| Injection frequency | Once weekly | Once weekly | Once weekly |
| Price point (ZA) | Mid-high (R2,500) | Entry-level | Premium |
Where Cagrisema Sits on the Efficacy Spectrum
Cagrisema occupies a clear middle position relative to the two reference pens above: stronger than semaglutide monotherapy by ~7.6 percentage points of weight loss, but ~5 percentage points behind tirzepatide on raw weight-loss percentage. The distinct mechanism (amylin co-agonism) suits patients who tolerated semaglutide poorly or hit a plateau around 6–10% loss. The matched ~7-day half-lives of both peptides allow synchronised once-weekly dosing without the staggering required by separate injections (Enebo et al., Lancet 202101751-7/fulltext)).
Verdict
Choose semaglutide-only when you are new to GLP-1s, budget-conscious, or want the gentlest tolerability ramp. In contrast, choose Cagrisema if you have stalled on semaglutide, need combined HbA1c and weight outcomes, or want amylin-driven satiety that GLP-1 alone does not replicate. Choose tirzepatide when maximum weight loss is the sole priority and cost is secondary.
How to Buy the Body Pharm Cagrisema 12 Pen in South Africa
Buying the Cagrisema pen through Beskinny.store takes five steps: confirm clinical eligibility, add the pen to your cart at R2,500, check out, receive cold-chain delivery, and inspect the package on arrival. As of this article’s June 2024 publication date, 25 units are in stock and shipping is available nationwide via Beskinny — South Africa’s premium weight loss store. Confirm current price and stock on the product page before ordering, as both can change.
A common concern is the legal grey area: Cagrisema is not SAHPRA-registered, and clinician oversight is essential for managing prescription compliance and safety.
Step 1: Confirm Eligibility With a Healthcare Provider
Semaglutide is a Schedule 4 medicine in South Africa (SAHPRA), and Cagrisema is contraindicated in patients with a personal or family history of medullary thyroid carcinoma or MEN 2 (FDA Wegovy PI). Book a consultation with your General Practitioner (GP) or endocrinologist before ordering. Bring your HbA1c, BMI, and current medication list.
Step 2: Add to Cart and Check Out
Visit the Body Pharm Cagrisema 12 Pen product page on Beskinny.store, select quantity, and proceed to checkout. Beskinny accepts Electronic Funds Transfer (EFT), instant card payments, and major South African gateways. When weighing alternatives, browse the full range of weight loss injections — the HD Semaglutide 10 Pen and Body Pharm Tirzepatide 30 Pen sit on either side of Cagrisema on the efficacy spectrum.
Step 3: Delivery and Cold-Chain Packaging
Orders ship in insulated, ice-pack-lined parcels to maintain the 2–8 °C range required for peptide pens. Metro deliveries (Johannesburg, Cape Town, Durban, Pretoria) typically arrive within 1–3 business days; outlying areas take 3–5.
Step 4: What’s in the Box
Each parcel contains one Body Pharm Cagrisema 12 mg multi-dose pen, the manufacturer’s dosing leaflet, and pen-handling instructions. Refrigerate the pen immediately on arrival, inspect the cartridge for cloudiness or particulates before first use, and never use a pen that arrived warm or damaged.
Storage, Handling & Pen Usage Tips
Refrigerate your Body Pharm Cagrisema 12 pen at 2–8 °C between doses, never freeze the device, and keep the pen in its original carton to protect the peptide from light. Once in use, the pen can sit at controlled room temperature (≤30 °C) for up to 28 days — guidance that mirrors the storage parameters published for Ozempic and Wegovy, since cold-chain integrity is critical for semaglutide and cagrilintide stability. Discard any pen that has been frozen, left above 30 °C, or exposed to direct sunlight.
A reasonable concern is travel and load-shedding. Plan ahead with an insulated cooler bag and rotate ice packs every 4–6 hours during outages.
Priming, Injecting and Disposing
Attach a fresh needle before every dose, prime the pen per the manufacturer’s leaflet until a small droplet appears at the tip, and rotate injection sites between abdomen, thigh, and upper arm. Drop used needles into a South African Bureau of Standards (SABS)-approved sharps container — never household waste — and never share a pen with another person, even a family member, due to bloodborne infection risk.
Missed Doses
When you miss a weekly injection and your next scheduled dose is more than 2 days away, inject as soon as you remember. When fewer than 2 days remain, skip the missed dose and resume your normal schedule — do not double up. Should you switch to alternatives like the HD Semaglutide 10 Pen or Body Pharm Tirzepatide 30 Pen, restart the manufacturer’s titration schedule rather than dose-matching.
Complementary Products to Stack With Cagrisema
Stacking is optional — Cagrisema works as a standalone protocol — but four evidence-supported adjuncts (NAD+, glutathione, GHK-Cu, and retatrutide) address the metabolic, structural, and oxidative pressures that accompany rapid weight loss. Always clear any combination with your prescribing clinician first, and avoid stimulants like clenbuterol alongside GLP-1 agents without medical supervision, as the cardiovascular load is poorly characterised.
A common concern is whether stacking adds risk without clear benefit. Each adjunct below targets a specific physiological consequence of rapid weight loss; none is essential.
Metabolic and Cellular Support
The Nicotinamide Adenine Dinucleotide (NAD+) supplement supports mitochondrial energy production and offsets the fatigue users report during the steep appetite suppression of weeks 4–12. Glutathione addresses oxidative stress, which rises during accelerated lipolysis as fat-stored toxins re-enter circulation.
Tissue Repair and Advanced Options
The Body Pharm GHK-Cu 50 Pen (a copper-binding peptide) supports skin elasticity and connective-tissue repair during a sustained caloric deficit — a common concern at the 15%+ body-weight loss range seen in SCALE-NEXT01548-3/fulltext) data. For experienced users who plateau on dual agonism, HD Labs Retatrutide 32 offers a triple-agonist (GLP-1/GIP/glucagon) next step. The triple-agonist carries a less mature safety profile and is suitable only after a full Cagrisema cycle and clinician review.
Frequently Asked Questions About Cagrisema 12
How quickly will I see weight loss results with Cagrisema 12?
Expect early appetite suppression within 7–14 days, but visible scale movement typically begins around weeks 4–6 once you reach the 0.5 mg dose. Meaningful clinical weight loss — 5% or more of body weight — generally appears by weeks 8–12, with the SCALE-NEXT trial01548-3/fulltext) showing ~15.6% reduction at 32 weeks.
Can I use Cagrisema if I am already on metformin?
Generally yes — no pharmacokinetic interaction is documented between metformin and either semaglutide or cagrilintide, and the REDEFINE 2 trial includes participants on background antidiabetic therapy. Additive GI side effects are common, however, and your prescribing clinician should confirm dosing before you stack the two agents.
Is Cagrisema 12 the same as Ozempic?
No. Ozempic contains only semaglutide, while Cagrisema combines semaglutide with cagrilintide — a long-acting amylin analogue that suppresses glucagon and slows gastric emptying through a separate receptor pathway. For semaglutide-only therapy, see the HD Semaglutide 10 Pen; for stronger dual-agonism, the Body Pharm Tirzepatide 30 Pen is an alternative.
How many injections are in one pen?
The Body Pharm Cagrisema 12 pen contains 12 mg of total active peptide, sufficient for approximately 8–12 weekly injections depending on your dose tier (0.25 mg starter